 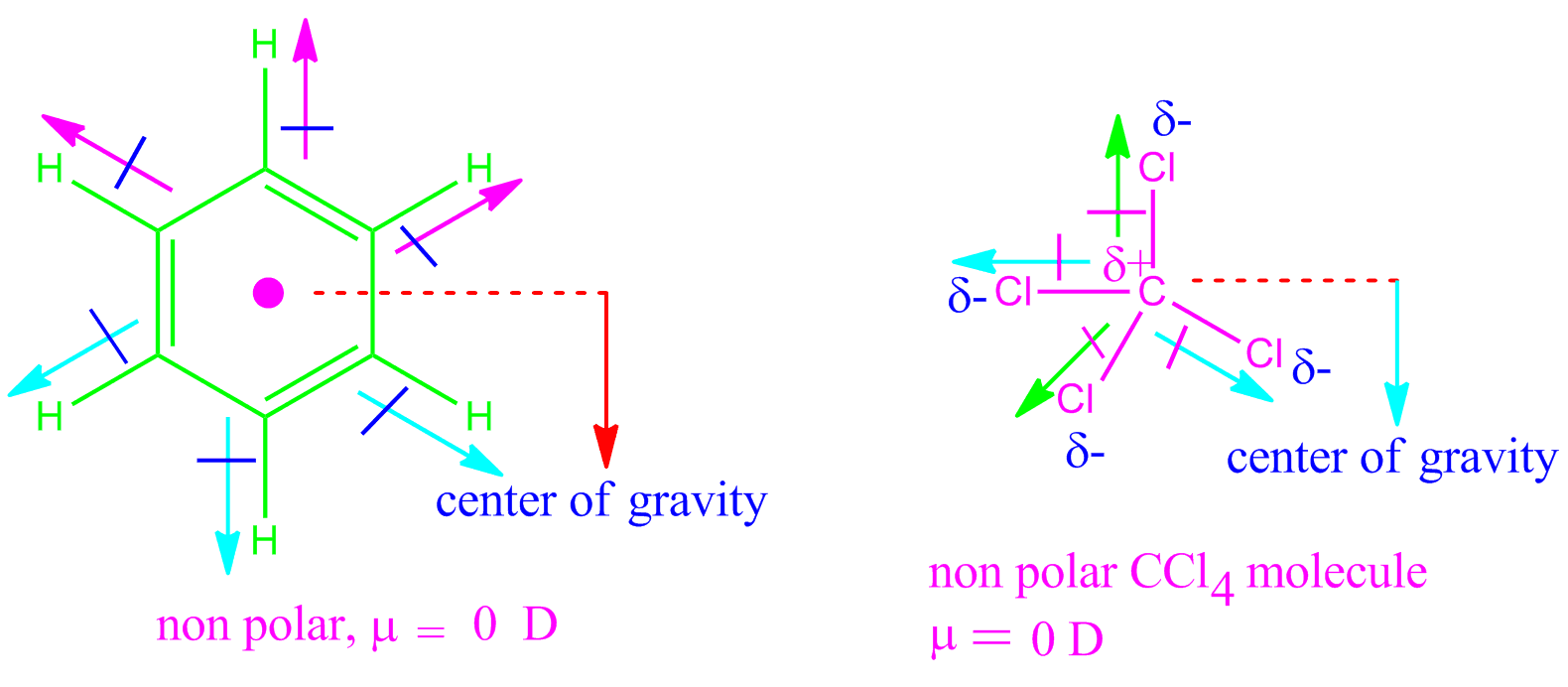 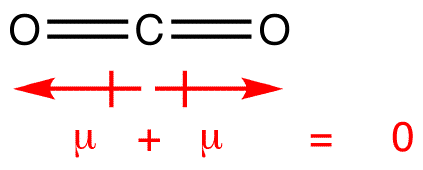
Medvedev, V.A.CODATA Key Values for Thermodynamics. KK Irikura "Experimental Vibrational Zero-Point Energies: Diatomic Molecules" J. 223 (1997) 59-98Ĭ Graham, DA Imrie, RE Raab "Measurement of the electric quadrupole moments of CO2, CO, N2, Cl2 and BF3" Mol. TN Olney, NM Cann, G Cooper, CE Brion, Absolute scale determination for photoabsorption spectra and the calculation of molecular properties using dipole sum-rules, Chem. Constants of Diatomic Molecules, Van Nostrand Reinhold Co., 1979 As a result, each bond dipole moments cancel out one another, resulting in zero net dipole moment. Structure of methane: Due to the tetrahedral structure, each bond pair is at an equal distance, indicating that they are symmetrically arranged. A water molecule, a commonly used example of polarity.Two charges are present with a negative charge in the middle (red shade), and a positive charge at the ends (blue shade). Herzberg, G., Molecular Spectra and Molecular Structure. The shape of methane CH 4 is tetrahedral. Please address comments about this page to Stern,RH Gammon, ME Lesk, RS Freund, WA Klemperer "Fine Structure and Dipole Moment of Metastable a 3Pi Carbon Monoxide" J. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. Because of this, CO2 strongly absorbs infrared radiation. However, some CO2 vibrations produce a structure with a molecular dipole. No inferences should be drawn on account of other sites being referenced, or not, from this page. Carbon dioxide doesn't have a molecular dipole in its ground state. We have provided these links to other web sites because they may have information that would be of interest to you. You are here: Experimental > One molecule all propertiesĮxperimental data for CO (Carbon monoxide)Ĭarbon monooxide Carbon monoxide Carbon oxide Carbon oxide (CO) Carbone Carbonic oxide Carbonio Kohlenmonoxid Kohlenoxyd Koolmonoxyde Monoxide NA 9202 Oxyde de carbone Įxperimental dipole measurement abbreviations: MW microwave DT Dielectric with Temperature variation DR Indirect (usually an upper limit) MB Molecular beamĬalculated electric quadrupole moments forĮlectric dipole polarizability (Å 3) alphaĬalculated electric dipole polarizability forīy selecting the following links, you may be leaving NIST webspace. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
